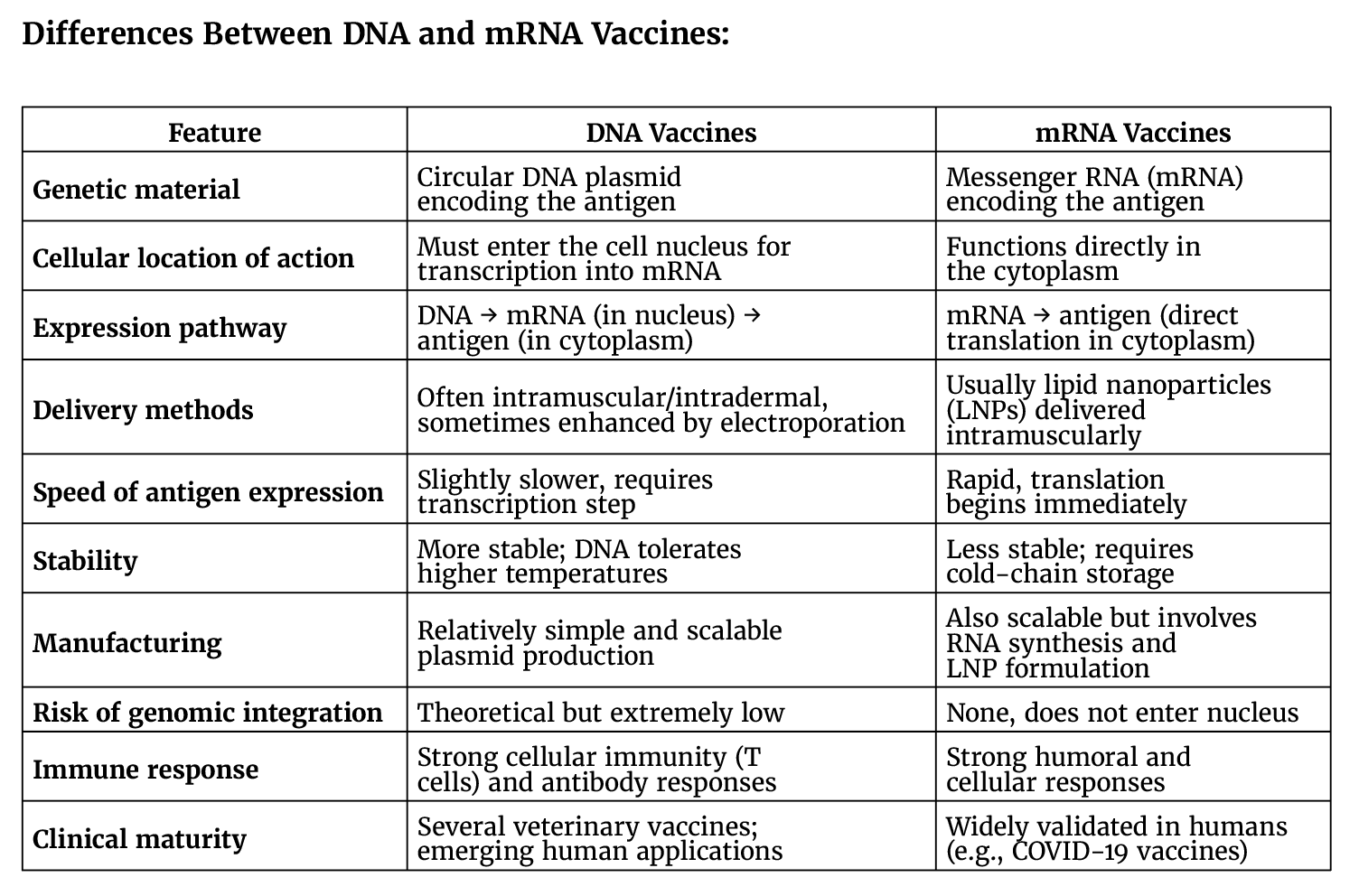
Introduction
Nucleic acid vaccines have recently gained considerable attention due to their promising results in clinical trials for the prevention and treatment of various diseases, including cancers and viral infections such as SARS-CoV-2. Compared with traditional protein- or virus-based vaccines, nucleic acid vaccines offer several advantages that make them one of the most promising platforms for disease prevention and therapeutic applications [3]. Unlike conventional vaccines, which often require lengthy production processes involving embryonated chicken eggs or cultured cells, nucleic acid vaccines can be designed and manufactured rapidly, allowing faster development and scalable production—an especially critical advantage in responding to emerging pandemics.
Deoxyribonucleic acid (DNA) vaccines have re-emerged as a versatile and scalable platform, driven by advances in synthetic biology and delivery technologies, positioning them as powerful tools in the post–mRNA vaccine era. Historically regarded as less potent than viral- or mRNA-based platforms, recent breakthroughs have substantially enhanced their immunogenicity, safety, and precision. These advances include the development of synthetic gene circuits, self-amplifying DNA (saDNA), and DNA-encoded monoclonal antibodies (DMAbs), enabling programmable antigen expression and more robust immune activation. Clinically, DNA vaccines are now expanding into a broad range of applications, from prevention of infectious diseases to therapeutic cancer immunotherapy and the treatment of immune-mediated disorders.
DNA vaccines: Structural conformations and implications for immunogenicity
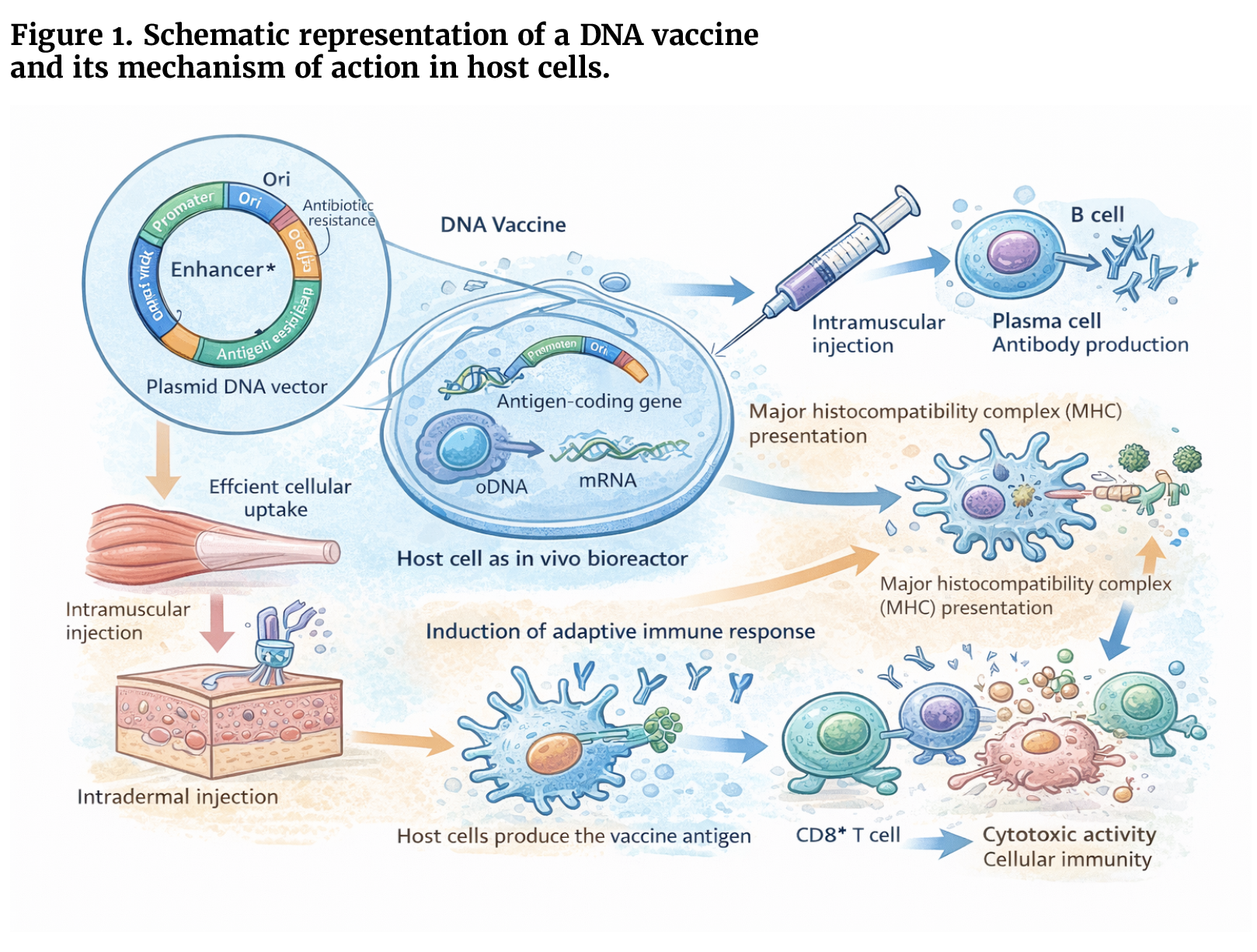
A DNA vaccine is typically generated by cloning a gene segment encoding a target antigen into a circular DNA vector—most commonly plasmid DNA or minicircle DNA constructs—which are designed for efficient expression in mammalian cells. Once engineered, the DNA construct is administered to the host through delivery routes such as intramuscular or intradermal injection, often combined with technologies that enhance cellular uptake, including electroporation or nanoparticle-based delivery systems.
Following administration, host cells—such as myocytes in intramuscular delivery or dermal antigen-presenting cells in intradermal administration—internalize the DNA and utilize their transcriptional and translational machinery to express the encoded antigen. In this process, host cells effectively function as in vivo bioreactors, producing the vaccine antigen directly within the body. The synthesized antigen is subsequently processed and presented through major histocompatibility complex (MHC) pathways, leading to activation of both humoral and cellular immune responses. This endogenous antigen production closely mimics natural infection, enabling DNA vaccines to stimulate robust CD4+ and CD8+ T-cell responses as well as antigen-specific antibody production (see Figure-1).
Current developments of DNA vaccines:
1. Improved delivery technologies
One of the historical limitations of DNA vaccines has been efficient delivery into host cells. Recent developments have significantly improved this through:
- Electroporation, which enhances cellular uptake of plasmid DNA and increases antigen expression.
- Needle-free jet injection systems and microneedle arrays.
- Nanoparticle and polymer-based delivery platforms that improve stability and targeting.
These technologies have markedly increased the immunogenicity of DNA vaccines in humans.
2. Synthetic biology and optimized plasmid design
Modern DNA vaccine platforms now incorporate several molecular innovations, including:
- Codon optimization to improve antigen expression.
- Synthetic promoters and regulatory elements to enhance transcription.
- Minicircle DNA vectors, which remove bacterial backbone sequences to improve safety and expression efficiency.
- Self-amplifying DNA (saDNA) constructs that increase antigen production.
These approaches make DNA vaccines more potent and precise.
3. DNA-encoded therapeutics
A major emerging field is the use of DNA to encode therapeutic proteins, including:
- DNA-encoded monoclonal antibodies (DMAbs)
- DNA-encoded cytokines and immunomodulators.
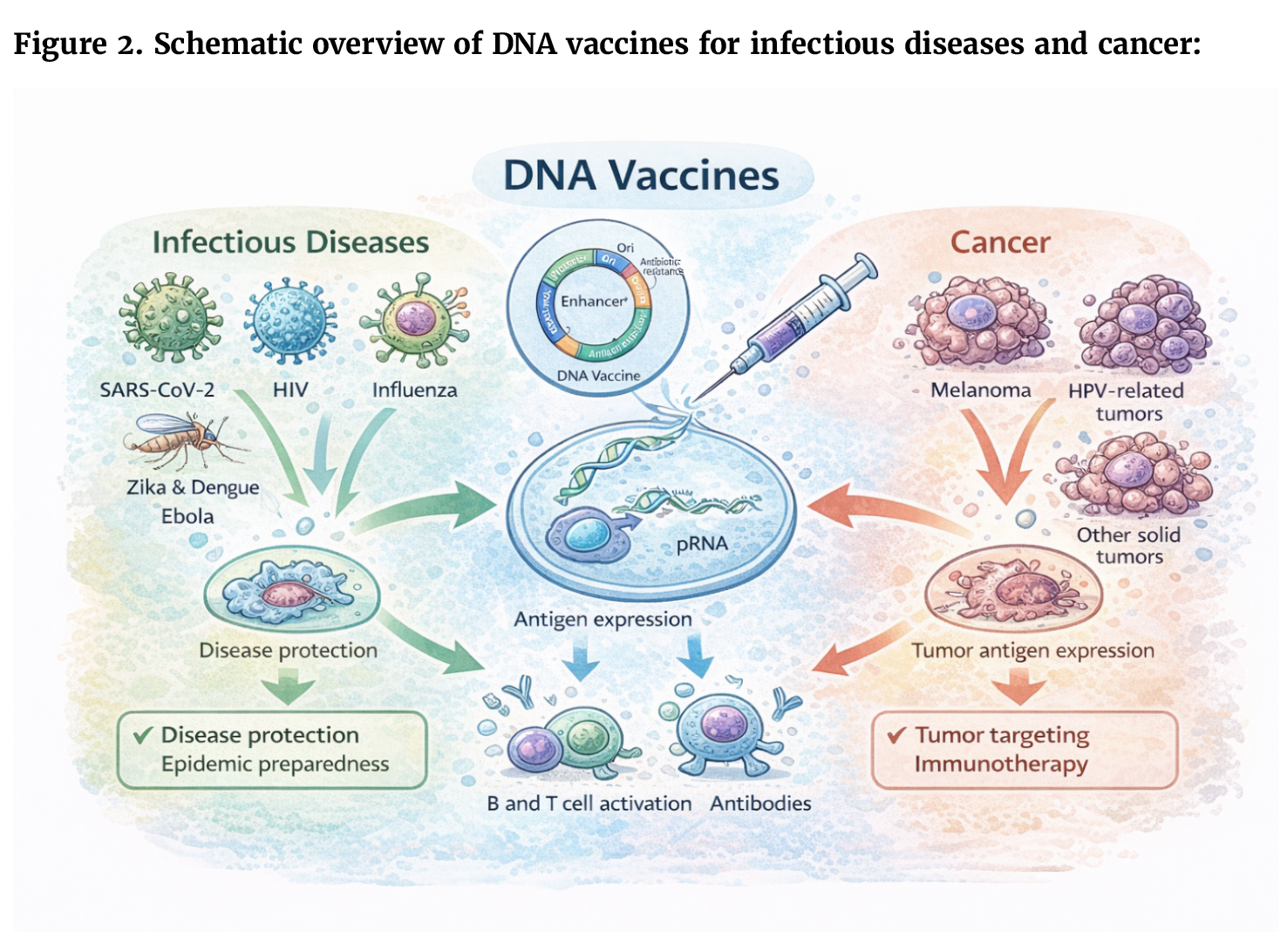
Current Landscape of DNA Vaccines in Infectious Diseases:
1. Licensed or Authorized DNA Vaccines:
Although most DNA vaccines remain investigational, a few important milestones demonstrate the platform’s clinical viability.
SARS-CoV-2:
- ZyCoV-D (India) – the first human DNA vaccine authorized for use (2021).
- Uses a plasmid DNA platform delivered intradermally with a needle-free injector.
- Demonstrated the feasibility of DNA vaccines for large-scale pandemic response.
This authorization marked a major milestone in nucleic-acid vaccinology and highlighted the speed and scalability of DNA vaccine manufacturing.
2. Major Viral Targets in Clinical Development
HIV
HIV has historically been one of the first targets for DNA vaccine technology, with early clinical trials beginning in the late 1990s. These trials established proof of concept for safety and immunogenicity, although achieving strong protective efficacy remains challenging.
Current research focuses on:
- Prime-boost regimens (DNA prime + viral vector boost)
- Mosaic antigen constructs to address viral diversity.
Influenza
DNA vaccines targeting influenza hemagglutinin or conserved viral proteins are under evaluation. These candidates aim to:
- Enable rapid pandemic vaccine production
- Potentially support development of universal influenza vaccines.
Zika Virus
Following the 2015–2016 outbreak, DNA vaccines rapidly entered clinical testing.
- Early candidates such as GLS-5700 (Inovio) progressed to human trials and demonstrated safety and immunogenicity.
Zika vaccine development became a model for rapid nucleic-acid vaccine design after genome sequencing of emerging pathogens.
SARS-CoV-2
Beyond ZyCoV-D, multiple DNA vaccine candidates were developed globally:
Examples:
- INO-4800 (Inovio)
- INO-4802, a pan-variant COVID-19 DNA vaccine candidate designed to target multiple variants simultaneously.
These efforts illustrated how DNA vaccines can be rapidly adapted during emerging pandemics.
Major Cancer Targets in DNA Vaccine Development:
In general, these are the key advantages of DNA Cancer Vaccines:
- Induction of strong CD8+ T-cell responses
- Ability to encode multiple tumor antigens
- Rapid and scalable manufacturing
- Good safety-profile in clinical trials.
HPV-related cancers:
The most advanced DNA cancer vaccines target cancers caused by human papillomavirus (HPV).
Example: VGX-3100
- DNA vaccine encoding HPV16 and HPV18 E6/E7 oncogenic proteins
- Targets cervical intraepithelial neoplasia (precancerous lesions)
- Evaluated in Phase III clinical trials and shown to induce regression of HPV-related lesions.
This program represents one of the most clinically advanced DNA vaccine approaches in oncology.
Melanoma:
Several DNA vaccines target melanoma antigens.
Example: SCIB1
- Plasmid DNA vaccine encoding melanoma antigens TRP-2 and gp100
- Designed to induce strong cytotoxic T-cell responses
- Evaluated in Phase I/II clinical trials.
These vaccines aim to stimulate immune recognition of melanoma cells expressing these antigens.
Breast cancer:
DNA vaccines targeting breast cancer antigens and neoantigens are under early clinical investigation.
Example:
- Poly-epitope DNA vaccines targeting tumor antigens in triple-negative breast cancer
- Early trials demonstrated strong immune responses and encouraging survival outcomes.
Lung cancer:
Some DNA vaccines encode multiple tumor antigens for non-small cell lung cancer (NSCLC).
Example:
- STEMVAC, a multi-antigen DNA vaccine targeting proteins involved in cancer stem cells and tumor progression.
Categories of DNA Cancer Vaccines:
1. Tumor-associated antigen vaccines
These vaccines encode shared cancer antigens found in many patients.
Common targets include:
- HER2/neu
- PSA (prostate-specific antigen)
- MUC1
- WT1
- HPV E6/E7
Advantages:
- Easier manufacturing
- Applicable to larger patient populations.
2. Neoantigen DNA vaccines
A newer approach involves personalized cancer vaccines.
These vaccines encode tumor-specific mutations unique to each patient, identified by tumor genome sequencing.
Advantages:
- Highly specific
- Reduced risk of targeting normal tissues.
This strategy is becoming central in precision oncology.
3. DNA-encoded immunotherapies
Another emerging strategy is DNA-encoded monoclonal antibodies or cytokines.
Examples:
- DNA-encoded checkpoint inhibitors
- DNA-encoded cytokines (IL-12)
This approach allows host cells to function as in vivo bioreactors producing therapeutic proteins.
4. Combination Strategies
Most cancer DNA vaccines are now being tested in combination therapies, such as:
- Checkpoint inhibitors (anti-PD-1, anti-CTLA-4)
- Radiotherapy
- Chemotherapy
- Oncolytic viruses
These combinations aim to overcome tumor immune evasion.
Conclusions:
DNA vaccines are emerging as a versatile platform with the potential to reshape modern immunotherapy and vaccinology. Their genetic flexibility allows rapid adaptation to newly emerging pathogens while also enabling the design of therapeutic strategies for complex diseases. Moving forward, progress will depend on integrating advances in systems biology, precision immunology, and translational research to refine antigen selection, improve delivery technologies, and better characterize immune responses.
The use of high-throughput genomic and immunological tools will facilitate the rational development of vaccines tailored to specific diseases and population needs. At the same time, precision immunology may support more individualized vaccination strategies by aligning vaccine constructs with the immune profile of each patient.
Equally important will be translating technological innovation into clinically feasible solutions. Scalable manufacturing processes, more efficient delivery systems, and reliable biomarkers of immune protection will be critical to advancing DNA vaccines from experimental platforms to widely applicable medical tools. With continued progress in these areas, DNA vaccines could become a powerful and adaptable approach for both preventive and therapeutic interventions across infectious diseases, cancer, and immune-mediated conditions.
Bibliography
- Neeli P, Chai D, Roy D, Prajapati S, Bonam SR. DNA Vaccines in the Post-mRNA Era: Engineering, Applications, and Emerging Innovations. Int J Mol Sci. 2025 Sep 7;26(17):8716. doi: 10.3390/ijms26178716.
- Wang C, Yuan F. A comprehensive comparison of DNA and RNA vaccines. Adv Drug Deliv Rev. 2024 Jul;210:115340. doi: 10.1016/j.addr.2024.115340.
- Amanpour S. The Rapid Development and Early Success of Covid 19 Vaccines Have Raised Hopes for Accelerating the Cancer Treatment Mechanism. Arch Razi Inst. 2021 Mar;76(1):1-6. doi: 10.22092/ari.2021.353761.1612.
- Bhojnagarwala PS, Jose J, Zhao S, Weiner DB. DNA-based immunotherapy for cancer: In vivo approaches for recalcitrant targets. Mol Ther. 2025 Jun 4;33(6):2719-2739. doi: 10.1016/j.ymthe.2025.04.008.
- Tiptiri-Kourpeti A, Spyridopoulou K, Pappa A, Chlichlia K. DNA vaccines to attack cancer: Strategies for improving immunogenicity and efficacy. Pharmacol Ther. 2016 Sep;165:32-49. doi: 10.1016/j.pharmthera.2016.05.004.
- Kaushik M, Madeswaraguptha P, Vanangamudi M, Surendran V, Ashique S, Islam A, Mojgani N, Hussain A. Recent progress, challenges (stability and in vivo distribution), and opportunities of DNA & mRNA vaccine in cancer immunotherapy. Adv Immunol. 2025;166:211-258. doi: 10.1016/bs.ai.2024.10.008.
- Sen A, Singh V, Ashique S, Jagriti, Biswas S, Islam A, Ehsan I, Mojgani N. DNA and mRNA vaccines: Significant therapeutic approach against cancer management. Adv Immunol. 2025;165:1-36. doi: 10.1016/bs.ai.2024.10.007.
- Pagliari S, Dema B, Sanchez-Martinez A, Montalvo Zurbia-Flores G, Rollier CS. DNA Vaccines: History, Molecular Mechanisms and Future Perspectives. J Mol Biol. 2023 Dec 1;435(23):168297. doi: 10.1016/j.jmb.2023.168297.
- Manickan E, Karem KL, Rouse BT. DNA Vaccines – A Modern Gimmick or a Boon to Vaccinology? Crit Rev Immunol. 2017;37(2-6):483-498. doi: 10.1615/CritRevImmunol.v37.i2-6.140.
- Silveira MM, Moreira GMSG, Mendonça M. DNA vaccines against COVID-19: Perspectives and challenges. Life Sci. 2021 Feb 15;267:118919. doi: 10.1016/j.lfs.2020.118919.
- Lopes A, Vandermeulen G, Préat V. Cancer DNA vaccines: current preclinical and clinical developments and future perspectives. J Exp Clin Cancer Res. 2019 Apr 5;38(1):146. doi: 10.1186/s13046-019-1154-7.
- Sheridan C. First COVID-19 DNA vaccine approved, others in hot pursuit. Nat Biotechnol. 2021 Dec;39(12):1479-1482. doi: 10.1038/d41587-021-00023-5.