Introduction
Streptococcus pneumoniae remains one of the most important causes of preventable illness and death worldwide, particularly among children under five years of age. The burden is disproportionately concentrated in low- and middle-income countries, where access to timely diagnosis, treatment, and vaccination may be limited. Pneumococcal disease encompasses a wide clinical spectrum, ranging from common infections such as acute otitis media and sinusitis to severe invasive conditions including pneumonia, bacteremia, and meningitis.
Before the widespread introduction of pneumococcal conjugate vaccines (PCVs), pneumococcal disease caused an estimated 14.5 million cases and approximately 735,000 deaths annually among HIV-uninfected children under five years of age. Over the past two decades, the integration of PCVs into national immunization programs has dramatically altered this landscape. Modeling studies suggest that by 2015, global pneumococcal cases in this age group had declined to about 3.7 million, with deaths reduced to approximately 294,000.
The global expansion of PCV programs represents one of the major successes of modern vaccination strategies. Between 2000 and 2015, pneumococcal mortality in children under five declined by roughly 51%, largely attributable to vaccine introduction and increasing coverage. Continued scale-up of pneumococcal vaccination is expected to have an even greater impact, with projections indicating that PCVs could prevent nearly two million additional deaths among young children between 2021 and 2030.
By 2024, pneumococcal conjugate vaccines had been incorporated into the routine immunization schedules of 162 of the 194 WHO Member States. Global coverage with the final PCV dose reached approximately 67%. Among the 157 countries reporting national data after introducing PCV, median coverage was substantially higher, at 89%, although wide variation persists across settings. In Gavi-eligible countries, where pneumococcal disease burden remains highest, 50 of 57 countries had introduced PCV by 2024, with last-dose coverage averaging around 75%.
Pneumococcal protein-polysaccharide conjugate vaccines:
The first PCVC to be widely implemented in infant immunization programs was the 7-valent PCV (PCV7), introduced in 2000. Its success in reducing invasive pneumococcal disease rapidly led to the development of higher-valency vaccines designed to broaden serotype coverage. Countries that initially adopted PCV7 later transitioned to expanded formulations, most commonly the 13-valent vaccine (PCV13-Pfizer), while others introduced alternative products such as the 10-valent vaccine (PCV10-GSK).
Over time, the pneumococcal vaccine landscape has continued to evolve. In 2019, a new 10-valent vaccine developed by the Serum Institute of India (PCV10-SII, Pneumosil®) received WHO prequalification, providing an additional option particularly relevant for low- and middle-income countries. Beyond these widely used formulations, several other conjugate vaccines containing between 7 and 20 serotypes have received regulatory authorization, including three products with expanded coverage beyond 13 serotypes.
The next generation of pneumococcal vaccines is already advancing through late-stage clinical development. Some candidates aim to include more than 20 serotypes, while others explore innovative strategies such as alternative conjugation technologies or the use of pneumococcal proteins as carrier antigens, with the goal of improving immunogenicity and broadening protection against pneumococcal disease.
Serological endpoints for assessing immunogenicity and non-inferiority of pneumococcal conjugate vaccines (PCVs):
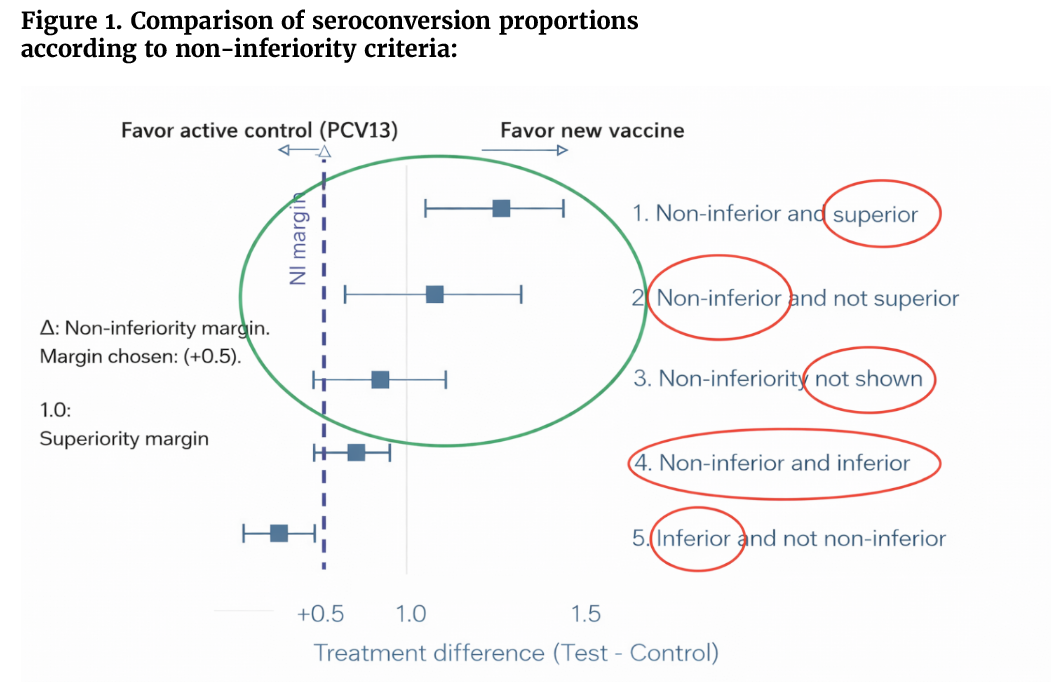
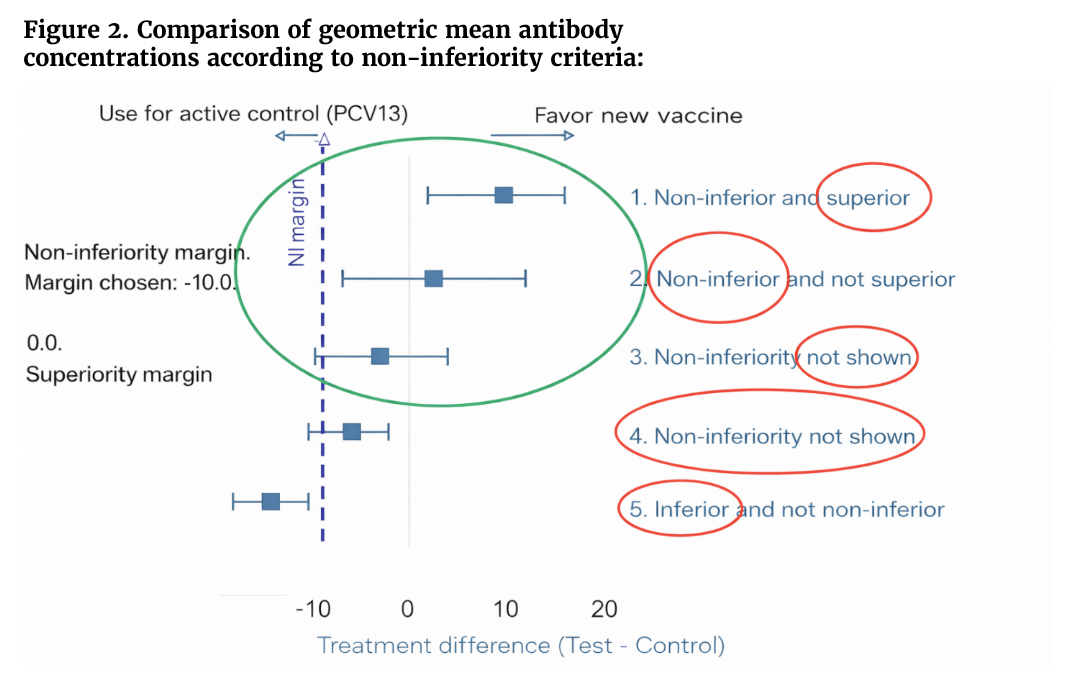
To facilitate the evaluation of new PCVs, the World Health Organization (WHO) has established immunological criteria designed to demonstrate non-inferiority compared with licensed reference vaccines. These criteria allow new formulations—including higher-valency vaccines—to be assessed without the need for large efficacy trials. The evaluation focuses primarily on two immunogenicity endpoints measured approximately four weeks after completion of the primary infant series.
The first endpoint is the proportion of vaccine recipients achieving serotype-specific immunoglobulin G (IgG) concentrations at or above 0.35 μg/mL, determined using the standardized WHO reference ELISA or an appropriately validated alternative assay. This threshold is widely considered a correlate of protection against vaccine-type invasive pneumococcal disease and is commonly referred to as the proportion of “Sero responders.”
The second endpoint is the geometric mean concentration (GMC) of serotype-specific IgG antibodies, which provides an overall measure of the magnitude of the immune response.
In comparative studies, non-inferiority is typically demonstrated when the candidate vaccine meets predefined statistical margins for both endpoints relative to the comparator vaccine. However, regulatory guidance recognizes that strict equivalence across all serotypes may not always be achievable. Consequently, if one or more serotypes fail to meet both criteria, satisfying at least one of the immunogenicity endpoints may still be considered acceptable for licensure, provided the overall immune profile remains comparable to that of the reference vaccine.
The 0.35 μg/mL threshold is based on pooled efficacy data from earlier trials of PCV7 and experimental higher-valency formulations, which demonstrated strong protection against vaccine-type invasive pneumococcal disease, corresponding to functional opsonophagocytic antibody titers (OPA) >1:8. Nevertheless, the relationship between lower serotype-specific GMCs and clinical protection remains incompletely understood. Importantly, these immunological benchmarks are intended to support aggregate assessments of non-inferiority across serotypes rather than to define absolute protection for each individual serotype. For this reason, trials must prespecify appropriate non-inferiority margins and implement statistical approaches that account for multiple serotype-specific comparisons (see Figures 1 and 2).
WHO recommendations for pneumococcal conjugate vaccines (PCVs) in children:
- At least three doses of a PCV are recommended in infancy to ensure adequate protection against pneumococcal disease.
- Vaccination schedules may follow either a 3+0 or a 2+1 regimen, depending on local or national epidemiology, programmatic considerations, and health system capacity.
- First dose: PCV should be administered starting at ≥6 weeks of age, typically aligned with routine infant immunization schedules.
- For the 2+1 schedule:
- The primary series consists of two doses, given at intervals of at least 8 weeks.
- A booster dose is recommended between 9 and 18 months of age to enhance and prolong immune protection.
- The 3+0 schedule consists of three primary doses in early infancy without a booster and may be preferred in settings where early protection against invasive pneumococcal disease is a priority.
- National immunization programs should select the schedule that best addresses local pneumococcal disease burden, transmission dynamics, and vaccine coverage patterns.
Catch-up pneumococcal vaccination in children
- In previously unvaccinated children aged 1 to 5 years, catch-up pneumococcal vaccination is recommended to provide protection against invasive pneumococcal disease.
- In children aged ≥24 months, a single dose of PCV can be administered as part of the catch-up strategy.
- In children aged 12–23 months, current evidence is insufficient to definitively determine whether one or two doses are required for optimal protection.
- Therefore, countries that adopt a single-dose catch-up strategy in this age group should carefully monitor vaccine impact and potential vaccine failures through surveillance systems.
- National immunization programs should consider local epidemiology, pneumococcal disease burden, and vaccine coverage levels when implementing catch-up vaccination strategies.
interchangeability of pneumococcal conjugate vaccines (PCVs)
- Currently, evidence on the interchangeability among all available PCVs remains limited.
- Once a PCV product has been introduced into a vaccination program, switching to another product is generally not recommended, unless there are substantial epidemiological, programmatic, or financial considerations.
- If the vaccination series cannot be completed with the same PCV product, the available PCV may be used to complete the schedule.
- Restarting a vaccination series with a different PCV is not recommended, including during the primary infant series.
- When a change in product is necessary, the replacement vaccine should be another who-prequalified PCV that has demonstrated adequate immunogenicity based on non-inferiority criteria.
Reduced-dose PCV schedules (1+1):
Countries seeking to reduce program costs or the number of injections in the infant immunization schedule may consider a 1 primary dose plus 1 booster (1p+1) PCV schedule as an off-label alternative to the standard 3-dose schedule, provided certain conditions are met. The best example currently is the United Kingdom.
Key requirements include:
- Strong population immunity in children under 5 years, demonstrated by:
- A well-established vaccination program with ≥80% coverage of the third dose during the previous 5 years,
- A recent multi-age vaccination campaign achieving ≥80% coverage in children under 5 years, or
- Low levels of vaccine-type pneumococcal carriage or disease, confirmed through reliable surveillance data.
- The capacity to deliver a booster dose between 9 and 18 months of age, with sustained vaccination coverage of ≥80% over the previous 5 years.
Before implementing a 1p+1 schedule, countries should also:
- Carefully evaluate the balance between potential cost savings and possible reductions in protection, and
- Ensure robust surveillance systems are in place to monitor invasive pneumococcal disease or pneumococcal carriage and detect any changes in vaccine-type disease.
Currently WHO-prequalified pneumococcal conjugate vaccines (PCVs):
Accumulating epidemiological evidence demonstrates that national PCV programs can substantially reduce vaccine-type invasive pneumococcal disease (IPD) and childhood pneumococcal pneumonia when implemented with high coverage. Historically, these public health gains have been achieved with WHO-prequalified vaccines such as PCV13 (Pfizer), PCV10 (GlaxoSmithKline), and PCV10 (Serum Institute of India), delivered through either a 3p+0 or a 2p+1 infant immunization schedule. More recently, the vaccine landscape has expanded with the introduction of additional higher-valency products, including the 14-valent pneumococcal conjugate vaccine (PCV14, Pneubevax®; Biological E.), which aims to broaden serotype coverage while maintaining strong immunogenicity. As newer PCVs become available, maintaining high vaccination coverage and appropriate scheduling remains critical to sustaining reductions in vaccine-type IPD, decreasing pneumococcal pneumonia in children, and strengthening population-level protection through herd effects.
Bibliography:
- WHO position paper: Pneumococcal conjugate vaccines in infants and children aged <5 years – September 2025. https://www.who.int/publications/i/item/who-wer10039-411-437.
- WHO: Pneumococcal Disease. https://www.who.int/teams/health-product-policy-and-standards/standards-and-specifications/norms-and-standards/vaccine-standardization/pneumococcal-disease.
- WHO Prequalification of Medical Products: Pneubevax 14®. https://extranet.who.int/prequal/vaccines/p/pneubevax-14r.
- von Gottberg A et al. Effects of vaccination on invasive pneumococcal disease in South Africa. N Engl J Med. 2014;371(20):1889–99.
- Malley R. Antibody and cell-mediated immunity to Streptococcus pneumoniae: implicationsfor vaccine development. J Mol Med (Berl). 2010;88:135–42. doi: 10.1007/s00109-009-0579-4.
- Francis JP et al. A longitudinal study of natural antibody development to pneumococcal surface protein A families 1 and 2 in Papua New Guinean Highland children: a cohort study. Pneumonia (Nathan). 2016;8:12. doi: 10.1186/s41479-016-0014-x.
- Feemster K et al. Implications of cross-reactivity and cross-protection for pneumococcal vaccine development. Vaccines (Basel). 2024;12(9):974. doi: 10.3390/vaccines12090974.
- Recommendations to assure the quality, safety and efficacy of pneumococcal conjugate vaccines. In: WHO Expert Committee on Biological Standardization, sixtieth report. Geneva: World Health Organization; 2013. Annex 3 (WHO Technical Report Series, No. 977; https://www.who.int/publications/m/item/pneumococcal-conjugate-vaccines-annex3-trs-977.
- Ngocho JS et al. Effectiveness of pneumococcal conjugate vaccines against invasive pneumococcal disease among children under five years of age in Africa: a systematic review. PLoS One. 2019;14:e0212295. doi: 10.1371/journal.pone.0212295.
- de Oliveira LH et al. Declines in pneumonia mortality following the introduction of pneumococcal conjugate vaccines in Latin American and Caribbean countries. Clin Infect Dis. 2020. doi: 10.1093/cid/ciaa614.
- Clarke E et al. Immunogenicity and safety of a novel ten-valent pneumococcal conjugate vaccine in healthy infants in the Gambia: a phase 3, randomised, double-blind, non-inferiority trial. Lancet Infect Dis. 2021;21:834-46. doi: 10.1016/S1473-3099(20)30735-0.
- Deloria Knoll M et al. Systematic review of the effect of pneumococcal conjugate vaccine dosing schedules on immunogenicity. Pediatr Infect Dis J. 2014;33 Suppl 2:S119–29. doi: 10.1097/INF.0000000000000079.
- Cohen O et al. Pneumococcal conjugate vaccine (PCV) review of impact evidence (PRIME). Geneva: World Health Organization; 2017. https://terrance.who.int/mediacentre/data/sage/SAGE_Docs_Ppt_Oct2017/9_session_PCV/Oct2019_session9_PCV_PRIMEsummary.pdf.
- Bertran M et al. Invasive pneumococcal disease 3 years after introduction of a reduced 1 + 1 infant 13-valent pneumococcal conjugate vaccine immunization schedule in England: a prospective national observational surveillance study. Lancet Infect Dis. 2024;24:546–56. doi: 10.1016/S1473-3099(23)00706-5.